 |
GLP-1 drugs are incredibly effective. But not unconditionally and not for everyone.
Genes might partly explain why.
Average weight loss in major trials of GLP-1 medications lands around 15%. But about one-third of people lose less than 5% of their initial body weight, and nearly two-thirds lose less than 10%.
A new study set out to test whether genetic factors might help explain these wide-ranging responses. It found that people with variations in certain genes—genes that encode for the very receptors that GLP-1-based medications target—lost more weight during treatment, but also were more likely to experience nausea and vomiting.
Today's newsletter will explore some of the implications for weight-loss treatments, and speculate on whether the "side effects" are a bug or a feature of why they work.
|
|
|
You just missed this in your inbox
Every week, Dr. Rhonda Patrick and the FoundMyFitness team distill the latest research into clear, actionable insights on health, longevity, and performance, delivered free to your inbox.
Genetic variation influences weight loss
Researchers conducted a large genome-wide association study, or GWAS, in 15,237 adults from the 23andMe research cohort who reported taking one or more GLP-1-based medications, including Ozempic, Wegovy, compounded semaglutide, Mounjaro, Zepbound, or compounded tirzepatide. A GWAS is essentially a large-scale scan across the genome to look for genetic variants associated with a measurable trait or outcome—in this case, weight loss and side effects while using GLP-1 drugs.
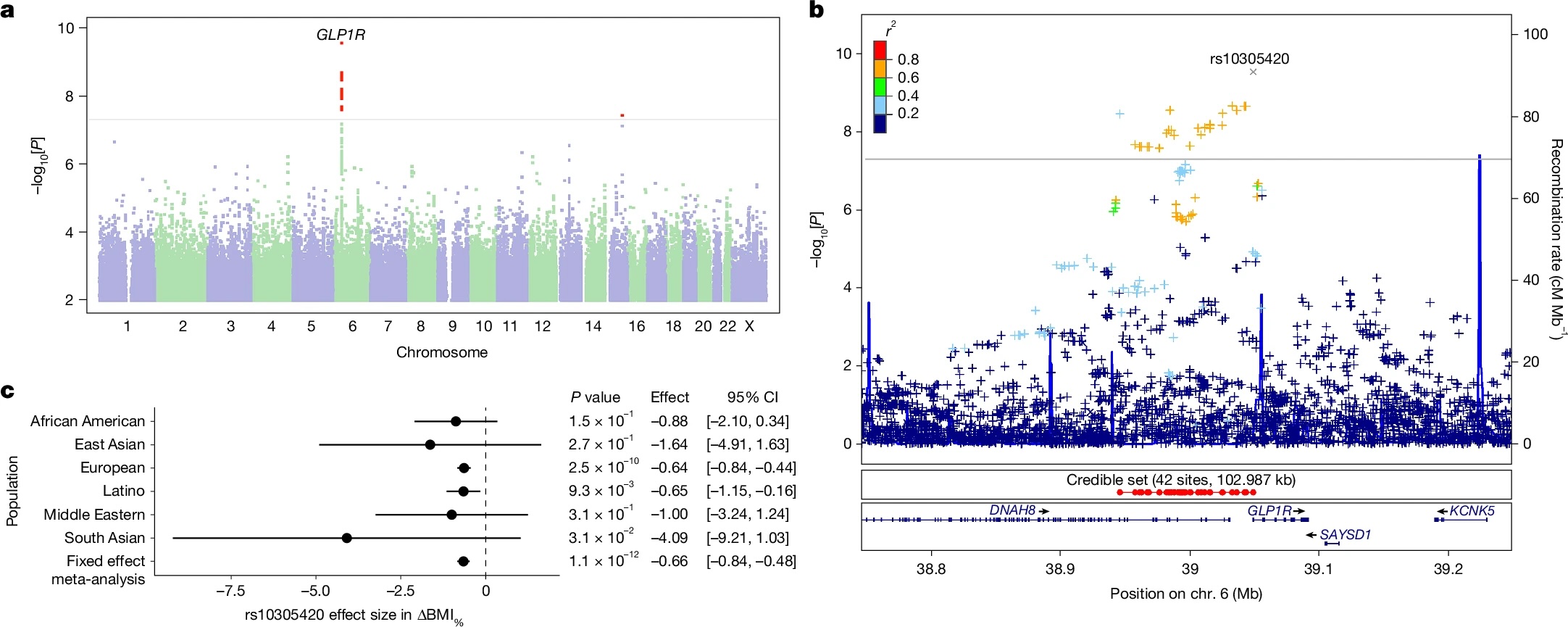
A common mutation was found in the gene encoding the GLP-1 receptor (GLP1R), which is the direct target of semaglutide and one of tirzepatide's targets.
Each copy of the T variant was associated with an additional 0.64% reduction in BMI, equivalent to about 1.7 pounds/0.8 kilograms of extra weight loss per allele. The effect was additive, meaning one copy helped a little and two copies helped more—someone inheriting two copies of the T variant would be expected to lose about 1.28% more BMI, or roughly 3.3 pounds/1.5 kilograms more weight, than someone with two C alleles.
This genetic variant is called rs10305420. It changes one DNA letter, from C to T, and that small change swaps the seventh building block of the protein from proline to leucine.
The authors propose that replacing proline with leucine may matter because leucine is more hydrophobic (avoiding interaction with water). That could stabilize the peptide, improve trafficking of the receptor through the cell, and ultimately increase how much GLP-1 receptor reaches the cell surface. This would increase the opportunity for semaglutide or tirzepatide to bind and signal. That mechanism is still speculative, but it is biologically plausible.
The effect also appeared somewhat stronger for tirzepatide than semaglutide. When the researchers looked at the drugs separately, the estimated effect of rs10305420 on BMI change was about 0.95% with tirzepatide versus 0.51% with semaglutide.
|
|
Genes predict side effects too
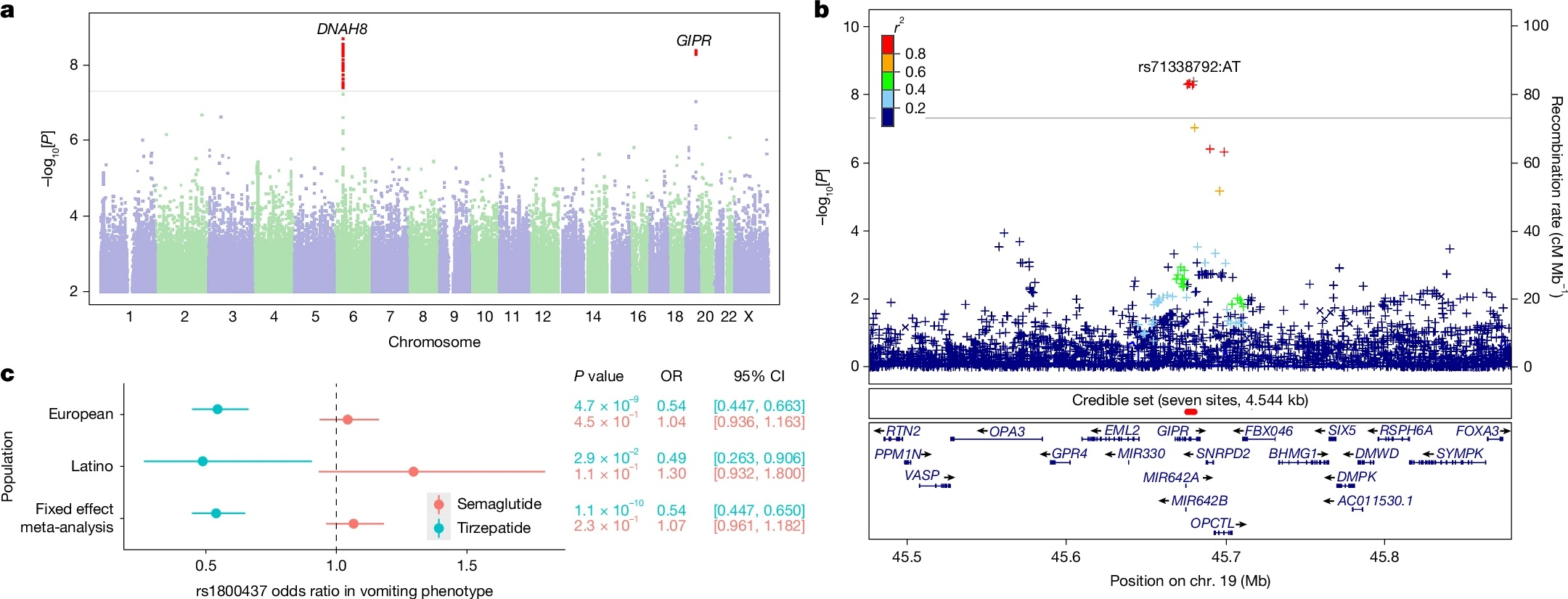
The researchers identified two strong signals in the GLP1R region itself that were associated with side effects in people using the drugs:
These two variants were not the same as the weight-loss coding variant, but they are part of the same broader genetic signal, and there was a high probability that the weight loss and side-effect signals overlapped, which led the authors to infer that the same genetic profile linked to greater weight loss may also increase the likelihood of nausea and vomiting.
In other words, the biology that makes the drug work better may also make it harder to tolerate.
That theme became even more interesting with tirzepatide, because tirzepatide is not just a GLP-1 receptor agonist—it is a dual receptor agonist that targets both GLP1 and gastric inhibitory polypeptide (GIP) receptors. The investigators found another association, this time in the gene encoding the GIP receptor (GIPR). Here, people carrying the variant had an approximately 83% higher odds of vomiting, but only if they were using tirzepatide, not semaglutide, and the variant did not seem to influence weight loss efficacy itself. This variant is known as rs1800437.
I think the authors’ mechanistic explanation is a good one. This variant has previously been characterized as a partial loss-of-function variant. If that dampens GIP receptor signaling, then it may reduce tirzepatide’s ability to counterbalance the nausea-inducing effects of GLP-1 signaling. This idea fits with preclinical work suggesting GIP receptor activation can blunt aversive effects like nausea and vomiting triggered by GLP-1 receptor activation.
So in someone carrying that GIPR variant, tirzepatide may behave more like a “less buffered” GLP-1 drug from the standpoint of tolerability.
People who carried both risk alleles at both the GLP1R and GIPR had a nearly 15-fold increase in the risk of vomiting compared with people carrying non-risk variants. That is a striking number. It does not mean those people are guaranteed to vomit, but it does illustrate the basic principle that gene variants can stack, and when they do, tolerability may shift dramatically.
|
|
| |
I've discussed the evidence on GLP-1 medications, my views on whether these drugs might have benefits beyond weight loss, and their potential side effects in a few of my recent Q&A episodes.
Q&A #66 (1/4/25)
-
06:58 — Could GLP-1 drugs help conditions like osteoarthritis or rheumatoid arthritis?
-
11:14 — Do GLP-1 receptor agonists offer cardiovascular benefits beyond weight loss alone?
-
13:02 — What is the early evidence for GLP-1 drugs in Alzheimer’s and Parkinson’s disease?
-
18:24 — What does the research suggest about GLP-1 drugs and obesity-related cancer risk?
-
22:15 — What are the potential bone-density risks during weight loss on GLP-1 drugs?
-
24:10 — Can resistance training help protect bone during GLP-1-associated weight loss?
Q&A #79 (3/14/26)
-
03:51 — How do semaglutide, tirzepatide, and newer triple-agonist drugs differ?
-
1:00:54 — How does GLP-1 affect insulin secretion, gastric emptying, and appetite?
-
1:06:00 — What are the main concerns about muscle loss, gallstones, and pancreatitis with rapid weight loss?
-
1:07:09 — Is microdosing GLP-1 drugs for longevity plausible, or still mostly speculative?
-
1:07:40 — Can GLP-1 drugs help reduce visceral fat?
-
1:10:28 — Why is weight regain so common after stopping GLP-1 drugs?
|
| |
|
Non-genetic predictors of weight loss
Several non-genetic predictors also shaped responses:
Women lost more BMI than men overall. Tirzepatide produced greater weight loss than semaglutide. Older age predicted slightly less efficacy, with each additional 10 years associated with about a 0.5% smaller BMI reduction. Type 2 diabetes was one of the strongest predictors of a weaker response, lowering expected BMI loss by about 2.8 percentage points.
The researchers then combined genetic and non-genetic predictors into response models, which did a moderately good job predicting weight loss, nausea, and vomiting risk.
This study makes a strong case that GLP-1 drug response is at least partly shaped by genetic factors. Variants in GLP1R may influence how much weight someone loses. Variants in GLP1R and GIPR may influence whether that person experiences nausea or vomiting, especially on tirzepatide. And because these variants lie in the genes encoding the therapeutic targets themselves, the findings are unusually (and satisfyingly) biologically coherent.
|
|
|
Predicted and observed changes in BMI in response to weight-loss medications in the prediction model. From: Ruth J. F. Loos (2026). doi: 10.1038/d41586-026-00905-1 |
| |
Are side effects a feature or a bug?
Part of what makes these weight-loss drugs so effective is that they don't act through a single pathway. GLP-1-based drugs cause people to eat less by acting on brain circuits involved in hunger, fullness (satiety), and food reward, while also engaging gut-brain signaling that slows gastric emptying and changes how "full" a meal feels. People taking these medications report less hunger, fewer cravings, and even a lower preference for high-fat, energy-dense foods.
But the same biology that makes food less compelling can also lead to gastrointestinal side effects—GLP-1 receptor agonists slow gastric motility and activate pathways in the brainstem that overlap with nausea and aversion. Roughly 50–75% of participants in trials of the leading GLP-1-based medications report some kind of gastrointestinal side effect, the most common of which is nausea, affecting about 25–50% of users, depending on the drug (it's higher with semaglutide compared to tirzepatide).
However, satiety and food aversion are not identical. Brain circuits that promote fullness can be separated from those driving food aversion (in animal models), and GLP-1 drugs reduce food intake even when the "aversion pathway" is inhibited. But the fact that feeling less hungry and feeling a little nauseous or sick "travel together" brings up an interesting… and slightly uncomfortable question:
Are some of these drugs' most notorious side effects partly tied to their success?
If taking Ozempic makes you feel fuller, slows gastric emptying, and in some cases causes nausea, it is reasonable to ask if eating less is driven by not wanting food because you're nauseous.
The data suggest this idea isn't entirely wrong, but probably overstated. People who experience gastrointestinal side effects sometimes lose slightly more weight than those who do not, suggesting side effects and efficacy are not totally separate. But the effect looks modest, not major:
In an analysis of 5 different weight-loss trials, only about 0.2–1 pound of semaglutide’s extra weight loss (compared to other treatments) was mediated by nausea or vomiting.1 In other analyses of semaglutide (compared with other GLP-1 receptor agonists or placebo), nausea and vomiting explained less than 0.2 pounds of the weight-loss advantage compared to other GLP-1 receptor agonists and less than 1% of extra weight loss compared to placebo.2, 3 An analysis of a weight-loss trial using tirzepatide concluded that gastrointestinal adverse events appeared to contribute slightly to weight reduction, but substantial weight loss still occurred regardless, suggesting nausea is not the main engine of the effect.4
So while side effects may modestly amplify weight loss in some people, especially early on, they do not appear to explain most of why these drugs work. The larger effect still seems to come from intended changes in appetite, satiety, cravings, and overall calorie intake.
If we return to today's study, perhaps the 25% of the variation in weight loss estimated to be accounted for by genes is related to nausea and vomiting side effects. That would line up neatly with a ~1 pound weight loss "advantage," in people carrying those specific risk variants, leaving the rest to be explained by non-genetic (and non side-effect-mediated) factors.
Importantly, this is not unique to GLP-1 drugs. The same pattern shows up again and again in nutrition and exercise research. There is no universal response. That idea is part of the reason I built the Genetic Report—to identify some of the most impactful genes and pair them with research-backed insights on nutrition and lifestyle, not as deterministic answers, but as better starting points.
(By the way, you can check your genotype for this particular polymorphism by uploading your raw genetic data in this free basic GLP-1 report on my website).
|
|
| | | |
| |  | |
| | | |
|
| |
Final thoughts
It’s tempting to look at results like these and conclude that we’re entering a true era of “precision medicine”—one where we can scan someone’s genome and predict exactly how they’ll respond to a given diet, training program, or weight-loss drug. But that conclusion is still premature.
Genes are clearly informative. In this study, genes explained roughly 25% of the variation in weight loss. But that also means the majority of the signal—about 75%—was driven by non-genetic factors. Environment, behavior, clinical context, adherence, baseline physiology… all of it still matters, and likely matters more.
So this is not a story where a single gene determines whether a drug will “work” for you. A more accurate way to think about it is that we are starting to sketch the early outlines of a map where receptor biology, genetics, lifestyle, and clinical characteristics all interact to shape outcomes. Some people land in the sweet spot. Others don’t. And we’re just beginning to understand why.
Regardless of where you land on GLP-1-based drugs, one thing is clear: they’re not going away. Their use is accelerating, and with that comes a growing need to understand why response varies so widely from one person to the next.
Not everyone is using weight-loss drugs. And not everyone should. But this study offers a credible first glimpse into a future where decisions around drug choice, dosing, and even side-effect risk might be informed by genotype. But that future isn’t here yet. We’re still early, and there’s a lot left to learn.
|
|
Warm regards
— Rhonda and the FoundMyFitness team
|
|
|
|
|
|